In Silico Evaluation of Flavonoids from Imperata cylindrica as Potential Antidiabetic Agents
Abstract
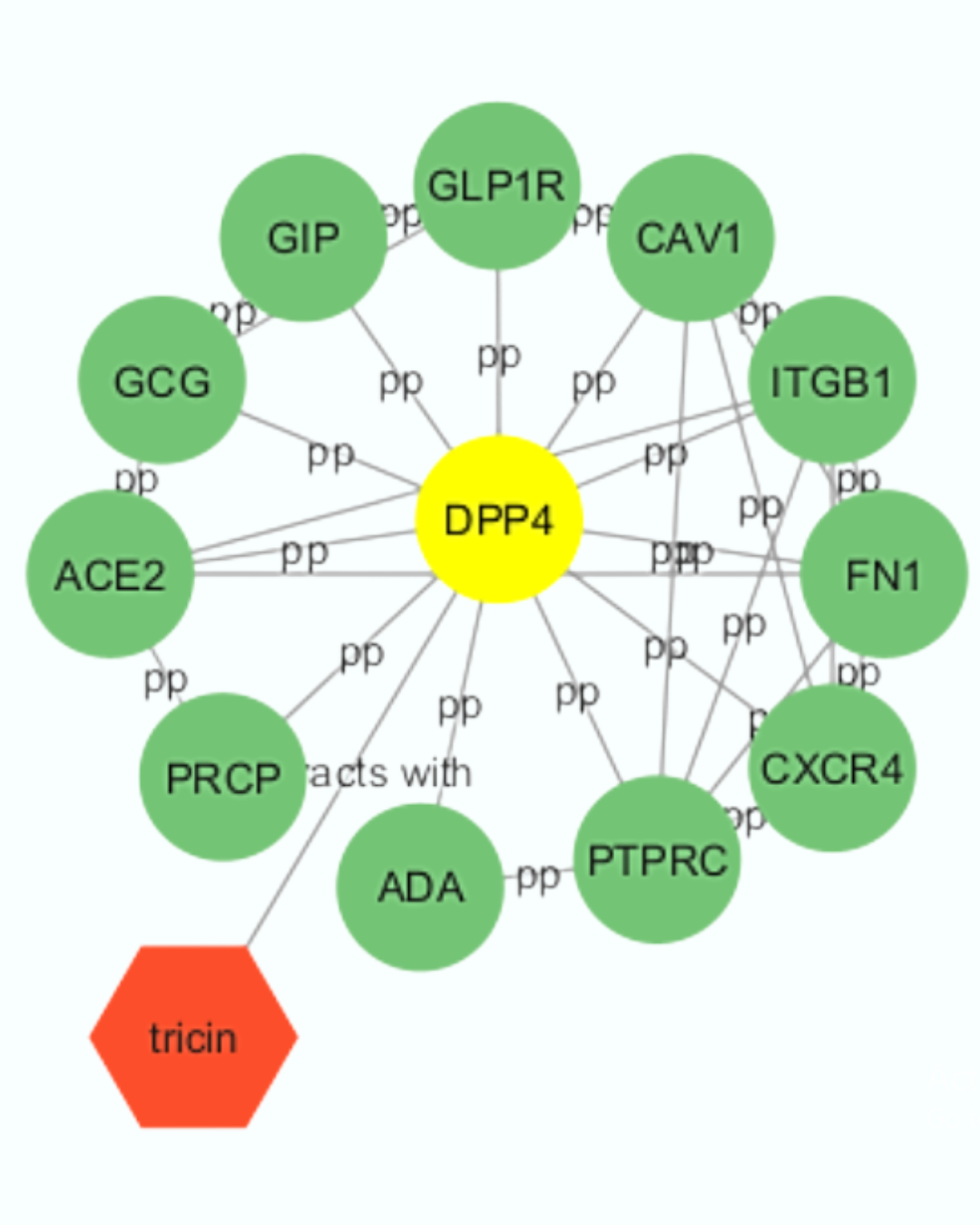
Diabetes mellitus is a metabolic disorder characterized by impaired insulin secretion and disrupted blood-glucose homeostasis. Novel therapies targeting the incretin system, such as dipeptidyl peptidase 4 (DPP-4) inhibitors, enhance insulin secretion, protect β-pancreatic cells, and restore glucose balance. Imperata cylindrica (reeds) has shown antidiabetic effects, including improved insulin release and reduced blood glucose, but its molecular mechanism remains unclear. This study investigated flavonoid compounds from I. cylindrica as potential DPP-4 inhibitors using molecular docking. The target protein (PDB: 6B1E) and drug-like flavonoids were evaluated via PyRx and visualized with Discovery Studio 2021. The results indicate that tricin binds strongly to DPP-4 (−8.4 kcal/mol) and forms stable interactions with residues ILEB102, ILEB76, and ILEB529. These findings suggest that tricin from I. cylindrica may inhibit DPP-4 and serve as a potential antidiabetic agent in silico.
Full text article
References
Afnani, M. R., Emilia, N. F., Damayanti, A. E., Rabbani, C. N., & Purnama, E. R. (2024). Potency of active compounds extract of soursop leaves (Annona muricata) as a candidate for cervical cancer drug in silico. E3S Web of Conferences, 513, 03005. https://doi.org/10.1051/e3sconf/202451303005
Anggarani, M. A., Purnomo, A. R., Ariyanto, D., Fajriyah, L., Wardani, M. K., Emilia, N. F., Liyana, W. A., Gustinia, N. S. N., Afnani, M. R., & Purnama, E. R. (2025). In silico Study of Ruminant Feed Wafer Formulation with Empon-Empon as Health Support Food. Tropical Journal of Natural Product Research, 9(8), 3695–3703. https://doi.org/10.26538/tjnpr/v9i8.28
Astuti, N. P. W., Manuaba, I. B. P., Jawi, I. M., Putra, A. A. B., Wiradana, P. A., Widhiantara, I. G., Permatasari, A. A. A. P., Ansori, A. N. M., & Kharisma, V. D. (2024). Phytoconstituents Analysis and Anti-Diabetic Potential of Sembung Leaf Extract (Blumea balsamifera L.) through Inhibition of NF-KB p65, GLP-1, and DPP-4 Proteins with In-Silico Approaches. Biomedical and Pharmacology Journal, 17(2), 1135–1148. https://doi.org/10.13005/bpj/2929
Chen, G., Seukep, A. J., & Guo, M. (2020). Recent Advances in Molecular Docking for the Research and Discovery of Potential Marine Drugs. Marine Drugs, 18(11), 545. https://doi.org/10.3390/md18110545
Deacon, C. F. (2020). Metabolism of GIP and the contribution of GIP to the glucose-lowering properties of DPP-4 inhibitors. Peptides, 125, 170196. https://doi.org/10.1016/j.peptides.2019.170196
Deshpande, R. R., Tiwari, A. P., Nyayanit, N., & Modak, M. (2020). In silico molecular docking analysis for repurposing therapeutics against multiple proteins from SARS-CoV-2. European Journal of Pharmacology, 886, 173430. https://doi.org/10.1016/j.ejphar.2020.173430
Elhady, S. S., Eltamany, E. E., Shaaban, A. E., Bagalagel, A. A., Muhammad, Y. A., El-Sayed, N. M., Ayyad, S. N., Ahmed, A. A. M., Elgawish, M. S., & Ahmed, S. A. (2020). Jaceidin Flavonoid Isolated from Chiliadenus montanus Attenuates Tumor Progression in Mice via VEGF Inhibition: In Vivo and In Silico Studies. Plants, 9(8), 1031. https://doi.org/10.3390/plants9081031
Florentin, M., Kostapanos, M. S., & Papazafiropoulou, A. K. (2022). Role of dipeptidyl peptidase 4 inhibitors in the new era of antidiabetic treatment. World Journal of Diabetes, 13(2), 85–96. https://doi.org/10.4239/wjd.v13.i2.85
Gu, H., Gao, J., Shen, Q., Gao, D., Wang, Q., Tangyu, M., & Mao, X. (2021). Dipeptidyl peptidase-IV inhibitory activity of millet protein peptides and the related mechanisms revealed by molecular docking. LWT, 138, 110587. https://doi.org/10.1016/j.lwt.2020.110587
Guzmán-Flores, J. M., Pérez-Vázquez, V., Martínez-Esquivias, F., Isiordia-Espinoza, M. A., & Viveros-Paredes, J. M. (2023). Molecular Docking Integrated with Network Pharmacology Explores the Therapeutic Mechanism of Cannabis sativa against Type 2 Diabetes. Current Issues in Molecular Biology, 45(9), 7228–7241. https://doi.org/10.3390/cimb45090457
Hira, T., Trakooncharoenvit, A., Taguchi, H., & Hara, H. (2021). Improvement of Glucose Tolerance by Food Factors Having Glucagon-Like Peptide-1 Releasing Activity. International Journal of Molecular Sciences, 22(12), 6623. https://doi.org/10.3390/ijms22126623
Jakhar, R., Dangi, M., Khichi, A., & Chhillar, A. K. (2020). Relevance of Molecular Docking Studies in Drug Designing. Current Bioinformatics, 15(4), 270–278. https://doi.org/10.2174/1574893615666191219094216
Ji, D., Xu, M., Udenigwe, C. C., & Agyei, D. (2020). Physicochemical characterisation, molecular docking, and drug-likeness evaluation of hypotensive peptides encrypted in flaxseed proteome. Current Research in Food Science, 3, 41–50. https://doi.org/10.1016/j.crfs.2020.03.001
Jia, C.-Y., Li, J.-Y., Hao, G.-F., & Yang, G.-F. (2020). A drug-likeness toolbox facilitates ADMET study in drug discovery. Drug Discovery Today, 25(1), 248–258. https://doi.org/10.1016/j.drudis.2019.10.014
July, Sauriasari, R., Syafhan, N. F., & Tahir, H. (2022). The Effect of Insulin Administration on Medication Adherence in Type 2 Diabetes Mellitus Patients with Neurological Complications. Jurnal Farmasi dan Ilmu Kefarmasian Indonesia, 9(3), 242–251. https://doi.org/10.20473/jfiki.v9i32022.242-251
Jung, Y.-K., & Shin, D. (2021). Imperata cylindrica: A Review of Phytochemistry, Pharmacology, and Industrial Applications. Molecules, 26(5), 1454. https://doi.org/10.3390/molecules26051454
Kato-Noguchi, H. (2022). Allelopathy and Allelochemicals of Imperata cylindrica as an Invasive Plant Species. Plants, 11(19), 2551. https://doi.org/10.3390/plants11192551
Nkonge, K. M., Nkonge, D. K., & Nkonge, T. N. (2023). Insulin Therapy for the Management of Diabetes Mellitus: A Narrative Review of Innovative Treatment Strategies. Diabetes Therapy, 14(11), 1801–1831. https://doi.org/10.1007/s13300-023-01468-4
Paggi, J. M., Pandit, A., & Dror, R. O. (2024). The Art and Science of Molecular Docking. Annual Review of Biochemistry, 93(1), 389–410. https://doi.org/10.1146/annurev-biochem-030222-120000
Pariyar, R., Bastola, T., Lee, D. H., & Seo, J. (2022). Neuroprotective Effects of the DPP4 Inhibitor Vildagliptin in In Vivo and In Vitro Models of Parkinson’s Disease. International Journal of Molecular Sciences, 23(4), 2388. https://doi.org/10.3390/ijms23042388
Pavlovicz, R. E., Park, H., & DiMaio, F. (2020). Efficient consideration of coordinated water molecules improves computational protein-protein and protein-ligand docking discrimination. PLOS Computational Biology, 16(9), e1008103. https://doi.org/10.1371/journal.pcbi.1008103
Rashid, R. S. M., Temurlu, S., Abourajab, A., Karsili, P., Dinleyici, M., Al-Khateeb, B., & Icil, H. (2023). Drug Repurposing of FDA Compounds against α-Glucosidase for the Treatment of Type 2 Diabetes: Insights from Molecular Docking and Molecular Dynamics Simulations. Pharmaceuticals, 16(4), 555. https://doi.org/10.3390/ph16040555
Raval, K., & Ganatra, T. (2022). Basics, types and applications of molecular docking: A review. IP International Journal of Comprehensive and Advanced Pharmacology, 7(1), 12–16. https://doi.org/10.18231/j.ijcaap.2022.003
Rendi, I. P., Maranata, G. J., Chaerunisa, H., Nugrahaeni, N., & Alfathonah, S. S. (2021). Molecular Docking of Compounds in Moringa oleifera Lam with Dipeptidyl Peptidase-4 Receptors as Antidiabetic Candidates. Jurnal Farmasi dan Ilmu Kefarmasian Indonesia, 8(3), 242. https://doi.org/10.20473/jfiki.v8i32021.242-249
Ruan, J.-Y., Cao, H.-N., Jiang, H.-Y., Li, H.-M., Hao, M.-M., Zhao, W., Zhang, Y., Han, Y., Zhang, Y., & Wang, T. (2022). Structural characterization of phenolic constituents from the rhizome of Imperata cylindrica var. major and their anti-inflammatory activity. Phytochemistry, 196, 113076. https://doi.org/10.1016/j.phytochem.2021.113076
Santos, C. M. M., Proença, C., Freitas, M., Araújo, A. N., Silva, A. M. S., & Fernandes, E. (2024). 2-Styrylchromones as inhibitors of α-amylase and α-glucosidase enzymes for the management of type 2 diabetes mellitus. Medicinal Chemistry Research, 33(4), 600–610. https://doi.org/10.1007/s00044-024-03200-8
Stanzione, F., Giangreco, I., & Cole, J. C. (2021). Use of molecular docking computational tools in drug discovery. Progress in Medicinal Chemistry, 60, 273–343. https://doi.org/10.1016/bs.pmch.2021.01.004
Stoian, A. P., Sachinidis, A., Stoica, R. A., Nikolic, D., Patti, A. M., & Rizvi, A. A. (2020). The efficacy and safety of dipeptidyl peptidase-4 inhibitors compared to other oral glucose-lowering medications in the treatment of type 2 diabetes. Metabolism, 109, 154295. https://doi.org/10.1016/j.metabol.2020.154295
Suhargo, L., Winarni, D., Fatimah, F., Kharisma, V. D., & Ansori, A. N. M. (2023). Antidiabetic Activity of Daun Wungu (Graptophyllum pictum L. Griff) Extract via Inhibition Mechanism of TNF-α, IL-6, and IL-8: Molecular Docking and Dynamic Study. Research Journal of Pharmacy and Technology, 2291–2296. https://doi.org/10.52711/0974-360X.2023.00376
Sun, Z.-G., Li, Z.-N., & Zhu, H.-L. (2020). The Research Progress of DPP-4 Inhibitors. Mini-Reviews in Medicinal Chemistry, 20(17), 1709–1718. https://doi.org/10.2174/1389557520666200628032507
Wang, X., Wang, Z.-Y., Zheng, J.-H., & Li, S. (2021). TCM network pharmacology: A new trend towards combining computational, experimental and clinical approaches. Chinese Journal of Natural Medicines, 19(1), 1–11. https://doi.org/10.1016/S1875-5364(21)60001-8
Wang, Z., Wang, J., Hu, J., Chen, Y., Dong, B., & Wang, Y. (2021). A comparative study of acarbose, vildagliptin and saxagliptin intended for better efficacy and safety on type 2 diabetes mellitus treatment. Life Sciences, 274, 119069. https://doi.org/10.1016/j.lfs.2021.119069
Yang, X., & Li, D. (2023). Tricin attenuates diabetic retinopathy by inhibiting oxidative stress and angiogenesis through regulating Sestrin2/Nrf2 signaling. Human & Experimental Toxicology, 42. https://doi.org/10.1177/09603271231171642
Yang, Y., Shi, C.-Y., Xie, J., Dai, J.-H., He, S.-L., & Tian, Y. (2020). Identification of Potential Dipeptidyl Peptidase (DPP)-IV Inhibitors among Moringa oleifera Phytochemicals by Virtual Screening, Molecular Docking Analysis, ADME/T-Based Prediction, and In Vitro Analyses. Molecules, 25(1), 189. https://doi.org/10.3390/molecules25010189
Zhang, B., Shen, Q., Chen, Y., Pan, R., Kuang, S., Liu, G., Sun, G., & Sun, X. (2017). Myricitrin Alleviates Oxidative Stress-induced Inflammation and Apoptosis and Protects Mice against Diabetic Cardiomyopathy. Scientific Reports, 7(1), 44239. https://doi.org/10.1038/srep44239
Zhu, W., Wang, Y., Niu, Y., Zhang, L., & Liu, Z. (2023). Current Trends and Challenges in Drug-Likeness Prediction: Are They Generalizable and Interpretable? Health Data Science, 3. https://doi.org/10.34133/hds.0098
Authors

This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. Copyright @2017. This is an open-access article distributed under the terms of the Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License (http://creativecommons.org/licenses/by-nc-sa/4.0/) which permits unrestricted non-commercial used, distribution and reproduction in any medium